Solutions for chiral separations & sample preparations
| Home |  | Products |

ChiralTek ChiralAMCE Column
for HPLC, SFC, and UPLC

ChiralTek ChiralCECD Column
for HPLC, SFC, and UPLC

ChiralTek ChiralCD Column
for HPLC, SFC, and UPLC
ChiralTek ChiralCE Column
for HPLC, SFC, and UPLC

ChiralTek ChiralAM Column
for HPLC, SFC, and UPLC

ChiralTek ChiralAMCD Column
for HPLC, SFC, and UPLC

ChiralTek Chiral Columns
for Preparative HPLC
ChiralTek manufactures the following three types of innovative products for chiral separations and sample preparations: (1) ChiralAM, ChiralCD, ChiralCE, ChiralAMCD, ChiralAMCE, and ChiralCECD series of novel chiral chromatography columns for HPLC, SFC, and UPLC; (2) HLE series of universal solid phase extraction (SPE) cartridges and UPE series of ultra-high performance SPE cartridges; and (3) the first type of no-cross-contamination SPE instrument PSPE and automated sample preparation instruments MFA and f-SPE.
1. High-enantioselective Chiral Columns for HPLC, SFC, and UPLC
A series of novel chemically-modified amyloses (AMs) or celluloses (CEs) were homogenously bonded onto high-quality porous silica particles in the amylose-immobilized ChiralAM columns or cellulose-immobilized ChiralCE columns via stable covalent bonds. Therefore, both standard and non-standard mobile phases can be used to optimize chiral separations in multi-mode chromatography. The ChiralCD series of chiral columns are new types of chemically-substituted cyclodextrin(CD)-bonded columns. The ChiralCD particles were synthesized by bonding a series of novel functional groups-substituted α-, β-, or γ-cyclodextrins onto surface of high-quality porous silica support by linking the spacer arms at the wider torus rims of the cyclodextrins. The ChiralAMCD, ChiralAMCE, and ChiralCECD columns are firstly created and produced by ChiralTek via immobilizing proprietary complex selectors containing two different chiral recognition moieties. They are the first type of commercial columns immobilizing the innovative complex selectors. Due to the co-operative functioning of the two different types of chiral recognition moieties of the novel complex selectors, the ChiralAMCD, ChiralAMCE, and ChiralCECD columns have exhibited excellent chiral recognition for a wide range of chiral compounds.
1.1. ChiralAMCD Columns
ChiralAMCD columns are the first type of chemically-modified amylose-appended cyclodextrin-bonded silica particles-packed chiral columns. The ChiralAMCD particles (shown in Figure (A)) were prepared through a specially-designed procedure by bonding the different functional groups-substituted amylose-appended cyclodextrin (AMCD) onto surface of high-quality porous silica (2 µm or 3µm for analytical columns, 5 µm or 10µm for preparative columns). The column contains a unique complex chiral selector with two recognition moieties: amylose and cyclodextrin.
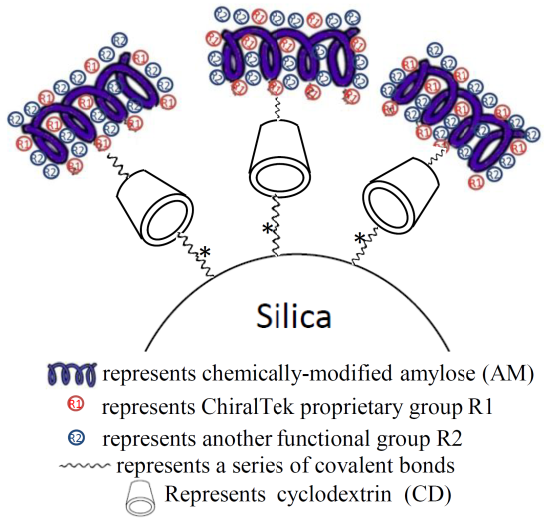
Figure (A). Schematic diagram of the bonded ChiralAMCD phase.
In the AMCD complex chiral selector, novel space structure with extra chiral recognition sites is formed between the amylose and cyclodextrin moieties. Due to the cooperative functioning of the amylose and cyclodextrin, the ChiralAMCD columns can provide different and generally better chiral separation abilities for a wider range of chiral compounds than other commercial amylose-based columns and cyclodextrin-based columns.
1.2. ChiralAMCE Columns
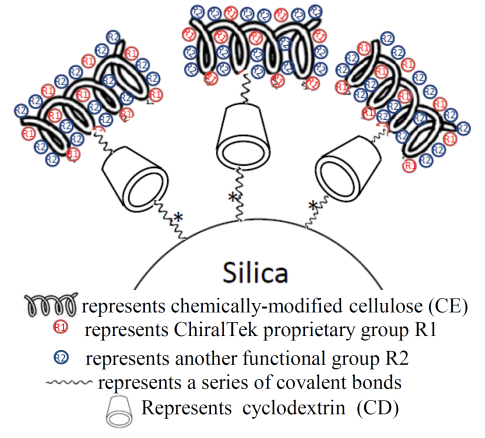
ChiralAMCE columns are the first and the only type of chemically-modified amylose-appended cellulose (AMCE)-bonded silica particles-packed chiral columns. The ChiralAMCE particles (shown in Figure (B)) contain a unique complex chiral selector AMCE with two recognition moieties: amylose and cellulose.
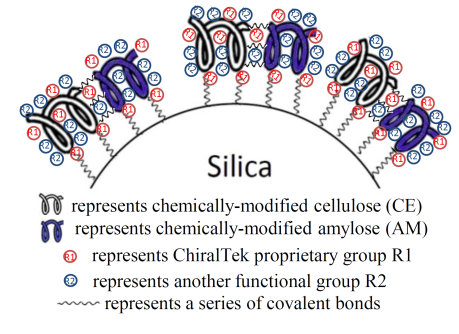
Figure (B). Schematic diagram of the bonded ChiralAMCE phase.
Other manufacturers’ chiral columns contain a single type of chiral selector (e.g., amylose, or cellulose, etc). However, the ChiralTek ChiralAMCE column contains both amylose and cellulose. Novel space structure with extra chiral recognition sites is formed between the amylose and cellulose moieties in the AMCE complex selector. This makes the AMCE selector possible to resolve more chiral compounds. Therefore, the ChiralAMCE columns can provide better chiral separations for a wider range of chiral compounds than other single chiral selector-based columns. Moreover, a single ChiralAMCE column can be used as two chiral columns: an amylose-based column and a cellulose-based column.
1.3. ChiralCECD Columns
The novel space structure with extra chiral recognition sites is formed between the cellulose and cyclodextrin moieties in the new CECD complex selector. It makes CECD able to recognize more chiral compounds. Due to this cooperative functioning of the cellulose and cyclodextrin, the ChiralCECD columns can provide different and generally better chiral separation abilities for a wider range of chiral compounds than other commercial cellulose-based columns and cyclodextrin-based columns.
1.4. ChiralAM Columns
ChiralAM columns are a new type of substituted-amylose-immobilized chiral columns. The ChiralAM particles were prepared through a specially-designed one-step reaction procedure by bonding the different functional groups-substituted amylose onto surface of high-quality porous silica support. Therefore, high column efficiency can be easily achieved on the ChiralAM columns. As shown in Figure (D), the amyloses were immobilized onto silica via stable chemical bonds.
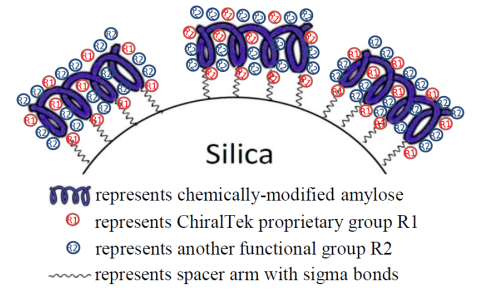
Figure (D). Schematic diagram of the bonded ChiralAM phase from ChiralTek.
Since the novel ChiralAM phases from ChiralTek have higher content of the bonded amyloses and contain more types of functional groups, the ChiralAM columns can provide different and generally better chiral separation abilities than other supplier’s amylose-bonded columns.
1.5. ChiralCE Columns
ChiralCE columns are a new type of cellulose-immobilized chiral columns. Several different functional groups-substituted celluloses have been successfully bonded onto the surface of the porous silica support. Therefore, the ChiralCE columns can be used under multiple modes of mobile phase conditions in UPLC, SFC, and HPLC. As shown in Figure (E), the chemically-modified celluloses were immobilized onto silica via stable chemical bonds. Both standard and non-standard mobile phases (e.g., chloroform and dichloromethane etc.) can be used to achieve optimized separation.
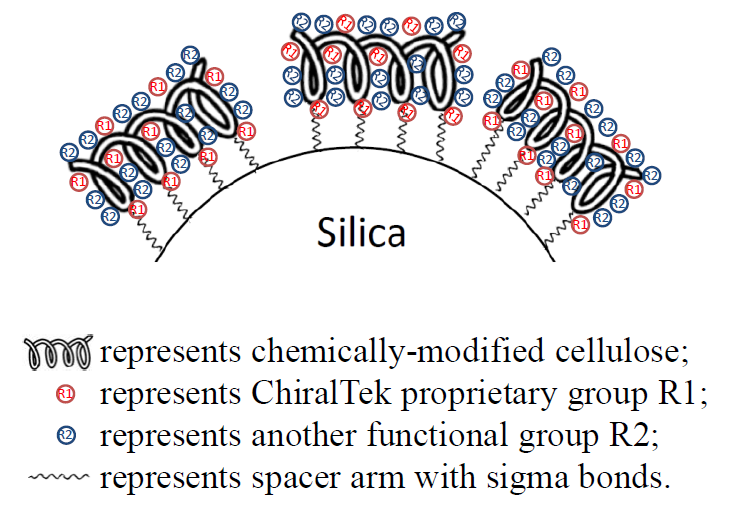
Figure (E). Schematic diagram of the bonded ChiralCE phase from ChiralTek.
Similar to ChiralAM phases, the new type of ChiralCE phases also have higher content of the bonded celluloses and contain more types of functional groups than others, the ChiralCE columns can provide different and sometimes better chiral separation abilities than other supplier’s cellulose-immobilized columns.
The chemical structures of the chiral selectors of ChiralAM, ChiralCE, ChiralAMCE phases are also different from other supplier’s phases. The general structure of the substituted-glucose unit of the chiral selector for ChiralAM, ChiralCE, and ChiralAMCE columns is showed in the following Figure (F).
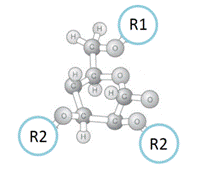
Figure (F). General structure of the substituted-glucose unit of the chiral selector for the ChiralAM, ChiralCE, and ChiralAMCE phases.
In the ChiralAM-1, ChiralCE-1, and ChiralAMCE-1 phases (Figure (G)), R2 is a Phenylcarbamate group. The ChiralAM-1, ChiralCE-1, and ChiralAMCE-1 columns can provide high column efficiency and excellent reducibility due to the contribution of the Phenylcarbamate group introduced into the bonded phase.
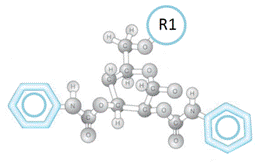
Figure (G). Typical structure of the substituted-glucose unit in the ChiralAM-1, ChiralCE-1, and ChiralAMCE-1 columns.
In the ChiralAM-2, ChiralCE-2, and ChiralAMCE-2 phases (as shown in Figure (H)), R2 is a 3,5-Dimethylphenylcarbamate group which was widely used in Phenomenex Lux Cellulose-1 column and Chiralcel OD, OD-H, OD-3, OD-RH, OD-3R, AD, AD-H, Chiralpak IA, and Chiralpak IB columns. Therefore, the ChiralCE-2 column is an ideal alternative column to the Phenomenex Lux Cellulose-1 column and Chiralcel OD, OD-H, OD-3, OD-RH, and OD-3R columns. The ChiralAM-2 columns are ideal alternative columns for Daicel Chiralpak IA, AD, and AD-H columns. Since a single ChiralAMCE-2 column can be used as both ChiralAM-2 and ChiralCE-2 columns, the ChiralAMCE-2 column is the ideal alternative column for the above-mentioned columns. Due to the extra chiral recognition functioning of the ChiralTek proprietary R1 group, ChiralAM-2, ChiralCE-2, and ChiralAMCE-2 can also provide some different and unique chiral separations for the solutes.
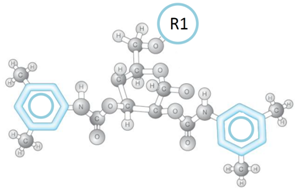
Figure (H). Typical structure of the substituted-glucose unit in the ChiralAM-2, ChiralCE-2, and ChiralAMCE-2 columns
In the ChiralAM-3, ChiralCE-3, and ChiralAMCE-3 phase (as shown in Figure (I)), R2 is 3-Chloro-4-methyl-phenylcarbamate group which was widely used in Phenomenex Lux Cellulose-2 column and Chiralcel OZ, OZ-H, OZ-3, OZ-RH, OZ-3R, and Chiralpak IC columns. Therefore, the ChiralCE-3 and ChiralAMCE-3 columns are ideal alternative columns to the Phenomenex Lux Cellulose-2 column and Chiralcel OZ, OZ-H, OZ-3, OZ-RH, OZ-3R, and Chiralpak IC columns. Due to the extra chiral recognition functioning of the ChiralTek proprietary R1 group, the ChiralAM-3, ChiralCE-3, and ChiralAMCE-3 can also provide some unique and better chiral separations for the solutes.
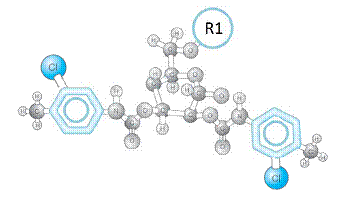
Figure (I). Typical structure the substituted-glucose unit in the ChiralAM-3, ChiralCE-3, and ChiralAMCE-3 columns
1.6. ChiralCD Columns
ChiralCD columns are a series of new types of chemically-substituted cyclodextrin-bonded silica particles-packed chiral columns. The ChiralCD particles were synthesized by bonding a series of different functional groups-substituted α-, β-, or γ-cyclodextrins onto surface of high-quality porous silica gel particles by linking the spacer arms at the wider torus rim of the cyclodextrins as shown in the following Figure (J). Due to the multiple functional groups, e.g., halogen groups, hydroxyl groups, aromatic rings, and cyclodextrin moiety etc., available in the bonded stationary phases, the ChiralCD columns can be used under multiple modes of mobile phase conditions in UPLC, SFC, and HPLC.
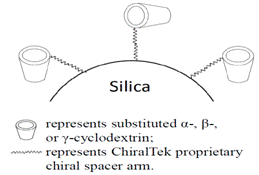
Figure (J). Schematic diagram of the CD-bonded ChiralCD phase.
At present, all the cyclodextrin-based chiral columns from other suppliers are prepared by anchoring cyclodextrin through the more reactive primary hydroxyl groups at the narrow torus rim of cycoldextrins. Since the ChiralCD columns are the first commercial chiral columns by anchoring cyclodextrins through the less reactive secondary hydroxyl at the wider torus rim of the cyclodextrins, the ChiralTek proprietary chiral spacer arms can provide extra unique steric interactions with solutes when entering the cyclodextrin cavities through the wider torus rim. Therefore, excellent separation selectivity can be achieved on the ChiralCD columns for separation a wide range of both chiral and non-chiral compounds. Since the ChiralCD phases from ChiralTek contain higher content of bonded cyclodextrins than other supplier’s cyclodextrin-bonded phases, the ChiralCD columns can exhibit much higher sample capacity. Due to the different chemical structure of the substituted cyclodextrins in ChiralCD phases, the new ChiralCD columns can provide different and generally better separation abilities than other supplier’s cyclodextrin-based columns.
2. Universal HLE Cartridges and Ultra-high Performance UPE cartridges for SPE
The Hydrophilic and Lipophilic Extraction (HLE) cartridges are a new type of universal solid phase extraction (SPE) cartridges manufactured by ChiralTek. The HLE packing materials were prepared by bonding multiple chemical function groups, including Diol, C18 (ODS), vinylpyrrolidone and divinylbenzene (HLB) etc., onto special type of porous composite materials. By using a ChiralTek proprietary packing approach, the HLE packing materials were then packed into kinds of polypropylene tubes with different sizes to produce many models of HLE cartridge products. Since the novel HLE cartridges contain multiple chemical function groups, they can be used under traditional normal and reversed phase elution conditions and modern gradient elution conditions to extract and to fractionate types of hydrophilic and lipophilic compounds from complicated samples. The HLE cartridges can also be used as direct replacement of HLB cartridges due to functioning of vinylpyrrolidone and divinylbenzene groups in the HLE particles.
The Ultra-high Performance Extraction (UPE) cartridges are a novel type of SPE cartridges manufactured by ChiralTek. The UPE particles were prepared through a specially-designed procedure by bonding C18 (ODS) and ChiralTek proprietary functional groups (R) onto high-quality porous spherical silica particles. Due to the unique co-operative functioning of the bonded C18 (ODS) and ChiralTek proprietary functional groups (R), the UPE cartridges exhibit excellent column efficiency, high reproducibility, high elution efficiency, and very high extraction recovery. The specially-designed synthetic procedure allows the UPE particles to contain ultra-high content of ODS functional group. This directly results in ultra-high sample capacity for the novel UPE cartridges containing ultra-small amount of packing materials. Therefore, the UPE cartridges only need ultra-small volume of organic solvents to achieve eluting and concentrating in one step. The novel UPE cartridges are idea choices for trace analysis when working with the PSPE, MFA & f-SPE instruments.
3. Novel Sample Preparation Instruments PSPE, MFA, and f-SPE
In order to maximize performance of the HLE and UPE cartridges, a series of innovative sample preparation instruments, including the first type of no-cross-contamination PSPE and novel flexible and versatile automated MFA & f-SPE systems, have been designed and successfully developed to perfectly work with the HLE and UPE cartridges. By using the ChiralTek novel SPE cartridges, the positive-pressure SPE instrument PSPE can conveniently provide innovative solutions for sample preparations without any cross-contamination. The multiple functional analyzer MFA and the fully-automated sample preparation station f-SPE can conveniently provide high-throughput automatic sample preparations when using the HLE and UPE cartridges.
3.1 The First Type of No-cross-contamination SPE Instrument PSPE
ChiralTek PSPE is the first instrument to be able to provide extraction of trace-level compounds without any cross-contamination when using ChiralTek novel SPE cartridges. The PSPE systems are designed for complicated biological and environmental samples. They are controlled by built-in single-chip microcomputer units (SCU) and driven by compact air-pumps with easy operations. They are the first type of compact, affordable, and cost-efficient commercial positive-pressure SPE systems which can completely eliminate cross-over contaminations. For small volume samples (0.1mL to 12mL), all the air-pump channels and fittings are not contacted with the samples and solvents during all processing procedures. The cross-over contaminations are therefore completely eliminated. This greatly improves the accuracy for kinds of trace-level analysis. There is no need to clean or wash the PSPE systems before and after extracting the small volume samples. This makes the PSPE systems very friendly to both environment and operators.
The PSPE systems are operated under 12 voltages. An external power supply with 100-240V input and 12V output (standard configuration) or a 12-volatge rechargeable storage battery (optional configuration) can be used to operate all the PSPE systems.
3.2. Automated Sample Preparation Instruments MFA and f-SPE
The MFA and f-SPE are manufactured by using a specially-designed flow-path-integration technique. Therefore, they are much smaller, robust, and more versatile than other commercial automated sample preparation instruments. ChiralTek MFA can easily work under both stepwise mode and timer mode. Stepwise mode is designed to automatically prepare solutions and clean tubing according to the step setting. The other timer mode can be used to deliver liquid or gas from one location to another according to the pre-set time points. When the two working modes are properly arranged in the methods, the MFA can be easily configured as solvent distributor shown in Figure [K1], feeder or sampler shown in Figure [K2] for chemical & biological reactors, natural product purifier shown in Figure [K3], or automated SPE as shown in Figure [K4]. Since it is equipped with a continuous pump with wide range of flow rate (0.1 to 90mL/min), the MFA exhibits great advantages with extra versatile applications such as, infusion analysis for LC-MS, step-gradient elution, and re-generation of SPE columns, etc.
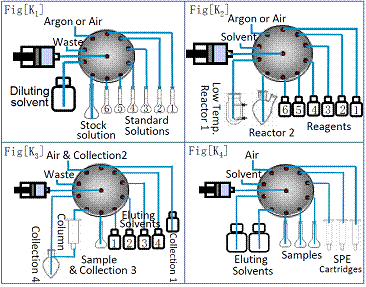
Figure (K). Typical configurations of MFA for multi-functional applications.
The MFA can also be used as portable equipment in fields when driven by an external uninterruptible power supply or 24-voltages storage battery.
ChiralTek f-SPE is a novel flexible and versatile platform for automatic sample preparations. It can be easily used as full-functional sample preparation station for automatic sample pre-treatments. The f-SPE can also be used as an autosampler for HPLC or MS system. When worked with ChiralTek HLE universal SPE cartridges, the f-SPE instrument can be used to conveniently achieve high efficient extraction and automatic online injection to HPLC or LCMS for full-automatic analysis of complicated samples. The f-SPE can work in offline mode and online mode. The f-SPE uses built-in methods to do offline SPE. It does not need a computer. Each step setting in the method controls action, flow rate, and volume. Methods can be easily edited and permanently saved. The operation of f-SPE involves only 7 buttons as shown in Figure (L). The f-SPE will automatically process samples according to the selected method.
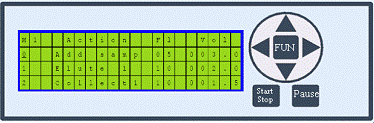
Figure (L). Offline control panel of the f-SPE station
In online mode, a computer with controlling software is applied to control f-SPE to perform full-functional sample preparation. There are more action functions in online mode than offline mode. Proper selection and combination of the action functions can easily perform the full-functional sample preparations. When worked with ChiralTek high-performance SPE cartridges. The f-SPE can automatically fulfill all necessary actions for solid phase extraction, such as conditioning of SPE columns, sample loading, cleaning tubing, drying sorbent, elution, fraction collection, and pre-concentration. It also can provide further online derivatization (optional) with two reagents and automatic online injection to HPLC or LCMS immediately after the extraction of the samples.
The f-SPE is a multiple functional platform for automatic sample preparations. It can provide not only faster sample pre-treatments, but also more precise results by reducing human error and improving laboratory safety and efficiency. The ChiralTek f-SPE can automatically process large number of different types of complicated samples when the system is properly configured with well-arranged online functions.
© ChiralTek Pte Ltd. All Rights Reserved.
192 Westwood Crescent, Singapore 648559

ChiralTek HLE Cartridges
for SPE

PSPE Instrument
without cross-contamination

MFA Multiple Functional Analyzer
for sample preparations

f-SPE Fully-automated Sample Preparation Station

Ultra-high performance UPE
Cartridges for SPE

